|
HOT
DIPPED GALVANIZING
|
|
|
|
|
|
 |
|
|
|
NEW!
- Hot dipped galvanized finish |
|
|
|
|
|
|
|
The
steel products displayed on this thumbnail page are now available
with a corrosion resistant finish applied to the steel after
fabrication.
Contact us for pricing! |
|
|
|
|
|
|
|
Hot
Dipped Galvanizing Corrosion Protection with Zinc
|
|
|
|
|
|
|
|
Steel
used to construct buildings, equipment, ornamental iron, bolts,
nuts and other small parts requires corrosion protection. The
proven
effectiveness of zinc in controlling corrosion makes it an ideal
choice
for protecting structural steel. Hot dipped galvanizing is generally
the
most effective method of applying zinc to steel. |
|
|
|
|
|
|
|
Why
Galvanizing Works |
|
|
|
|
|
|
|
The
excellent corrosion resistance of hot dipped galvanized steel
is due
to the two fold nature of the coating. The zinc coating provides
barrier
and cathodic protection to the underlying steel surface. |
|
|
|
|
 |
|
|
|
|
|
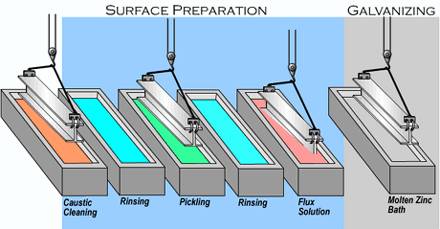
During
the Galvanizing process, steel reacts with molten zinc to form
a series of zinc-iron alloy layers which, together with the
outer zinc
layer, are metallurgically bonded to the steel surface. The
resulting
barrier coating has a very low corrosion rate (Figure 1) and,
because
of the hard alloy layers, is highly resistant to mechanical
damage.
Galvanized steel is much less susceptible to coating damage
from
handling than is painted steel. |
|
|
|
|
|
As
the name implies, hot dipped galvanizing requires complete immersion
of the work piece in molten zinc. This process ensures a uniform
coating
on all surfaces, even on those areas not normally accessible
to mechan-
ical applicators. |
|
|
|
|
 |
|
|
|
|
Zinc
corrodes preferentially to steel and provides cathodic protection
to
areas of bare steel (up to 1/4 inch wide) accidentally exposed
in service.
The sacrificial nature of zinc prevents the steel from corroding
adjacent
to the galvanized coating, thus preventing undercutting and
blistering of
the protective film. In comparison, exposed steel on painted
surfaces
will corrode immediately. The resulting rust occupies several
times the
volume of steel from which it was formed, causing the lifting
and
subsequent flaking of the adjacent paint film (Figure 2). |
|
|
|
CONTACT
US FOR PRICING!
|
|
|
|
|
|
|
|